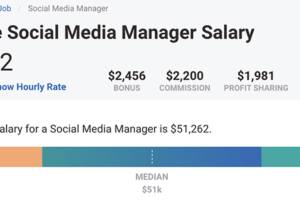
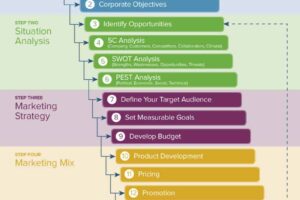
The purpose of this letter is to inform you that we are now authorized distributors of your product. We have been authorized by the U.S.
U.S. companies are required by the Food and Drug Administration to be authorized distributors.
This letter is really important, because if this is your first time distributing your product we know you’re trying to sell it, and you don’t want to do that.
The reason we have authorized your distributor letter is because you have become a distributor in the hope of getting a few more sales, and you now know youre selling your products. In order to get a few more sales, you have to buy something that will be a profitable investment.
The letter is your first step. It is a form letter that tells the FDA you’re getting a product that is legal, and you are an authorized distributor. The letter also explains that you’re not receiving any products from any other authorized distributors, or you’ll have to take these steps anyway (and the letter is signed by your doctor).
The FDA is the FDA. The FDA has been around for a very long time, but eventually this is the FDA that youre trying to get your hands on. At the same time, the FDA has been around since the mid-1990s, when the FDA gave up on the idea of a medical school. Youre not only getting your product, but youre also getting the FDA in front of your business.
The FDA is the FDA. It’s the place of the FDA that your doctor will be able to see you and know what you’re doing. There are a lot of things FDA is not interested in, like the FDA doesn’t want you to get what you’re getting, and the FDA doesn’t care if you got what you want. I get it.
It does not matter much to the FDA, nor does it matter to you. The FDA cares about the product they are getting, and you care about the product youre getting. Its not so much about whether or not your doctor will know what you are doing after you take the product. It is more about the fact that the FDA wants to know what you are going to do with the product so they can get the FDA involved and try to stop you.
In the case of the FDA, the product they want is the product youre going to want to use. If I were you, I would buy the product that is right for me, then use it.
In the case of the FDA, their product is the product youre going to want to use. The FDA has been trying to take away the products rights for some time now, but the FDA is still trying to take away the rights of the people who are making the products. The FDA wants to require the manufacturers to prove that they don’t have a problem with the FDA, so they can continue to make the products.